AI future impact on PCa: Bye-bye active surveillance for Gleason 6, hello AS for Gleason 8? Will the Gleason score and pathologist be replaced?
By Howard Wolinsky
Artificial intelligence seems to be changing everything. That includes prostate cancer.
Two issues in this area popped up in the Feb. 24 webinar on AI hosted by Active Surveillance Patients International (ASPI):
—Is active surveillance (AS) for patients with low-risk Gleason 6 prostate cancer going obsolete as AI emphasizes monitoring for more advanced cancers?
—Will AI replace the Gleason score and pathologists?
A couple of crystal ball gazers at the Feb. 24 AI webinar at ASPI indicated these changes could be in the offing in the years ahead.
In 13 years I’ve been on AS, I have seen rapid changes in technology and strategies. In 2010, we underwent annual biopsies; now, I haven’t had a biopsy in almost eight years. I was part of the first generation where magnetic resonance imaging was used, and likewise, the first gen for genomic testing.
AI is the next obvious game-changer,
(Dr. Freddie Hamdy)
In 2017, I was on a program with Dr. Freddy Hamdy, a top urologist from London and one of the leaders of the ProtecT study, who told me he thought AS should be phased out in patients with low-risk Gleason 6 disease who could live without intense monitoring of cancer at all and even not being diagnosed with the Big C.
Instead, patients with Gleason 7, he felt, were more ideal for surveillance, with them waiting to see if these slow-growing cancers suddenly became aggressive.
Breaking news--UK study reaffirms Active Management, surgery and radiation have similar survival rates
By Howard Wolinsky When British researchers launched the ProtecT study--a comparison of men who went on Active Monitoring (AM)—a grandaddy of today’s Active Surveillance—or were aggressively treated with surgery or radiation--they anticipated that survival curves eventually would separate, and those who underwent radical treatment would have a survival a…
These days, among patients with Gleason 3+4=7, a whopping 70% are considered to have “favorable” Gleasons that qualify them for surveillance.
But of those with low-risk Gleason 6, only 25% now go on AS nationally compared with 45% in the progressive MUSIC (Michigan Urological Surgeon Surgery Improvement Collaboration) in Michigan. (The Michigan program has 90%+ uptake of AS in men with Gleason 6 compared with 60% nationally.)
Michigan’s MUSIC hitmakers' record-breaking levels for AS with favorable intermediate-risk prostate cancer
By Howard Wolinsky Bob “Old Time Rock & Roll” and “Against the Wind” Seger and the Silver Bullet Band aren’t the only hitmakers to come out of the Mitten State. The Michigan Urological Surgery Improvement Collaborative (MUSIC), the pioneering group focused on improving urological care in Michigan, funded by Blue Cross Blue Shield of Michigan, has been h…
(Dr. Daniel Spratt)
Dr. Daniel Spratt, chair of radiation oncology at University Hospitals in Cleveland, told ASPI that he expects a transition into AS for men with Gleason 7s and even Gleason 8s. Yes, 8s.
“I think we've overtreated men with prostate cancer for decades. I think we'll find in a decade, Gleason 7s [and] I bet there are men with Gleason 8s on which we will do active surveillance [instead of aggressive treatment].”
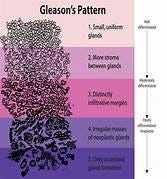
Spratt said Dr. Donald Gleason, who started the system carrying his name, never told doctors when or how these cellular irregularities should be treated. Rather, the Gleason system just classifies prostate cancer on its patterns.
(Dr. Gleason drew the original by hand. It first came into use in 1962.)
Spratt cited a study on treatment vs. watchful waiting--an earlier version of AS with no monitoring--(Scandinavian Prostate Cancer Group Study Number 4 (SPCG-4), a randomized trial of radical prostatectomy versus watchful waiting in men with localized prostate cancer) launched 30 years ago.
He said: “If you look at the guys with high-grade tumors—and this is again way more aggressive than modern-day high-grade tumors that were on watchful waiting—20 percent of them never develop metastatic disease or died of prostate cancer, and they've been followed for 30 years. And they have Gleason 8 to 10 tumors. So clearly there is biology that -- and if you were to tell anyone when I was in training, if you have Gleason 8 disease to watch it, you're insane. I'm not telling any of my patients at Gleason 8 to watch it, but what I'm saying is, I know I am overtreating men, and we need these tools to figure out who doesn't need treatment.”
I know a few men who are on AS even though they have been told they have Gleason 8s. They’re more are more afraid of potential damage to the quality of life [erectile disorder and incontinence] than they are of prostate cancer.
Meanwhile, thanks to changes in the ways men with suspected prostate cancer are evaluated with magnetic resonance scanning, the percentage of men diagnosed with prostate cancer has been dropping. Researchers from Weill Cornell reported in 2023 that between 2010 and 2019, the proportion of Gleason 6 patients had dropped significantly from 45% of all prostate cancer patients to 25%.
For most—not all of us—AS is better choice than active treatment, but it still can expose men to risks, such as potentially deadly and disabling sepsis and other infections from transrectal biopsies, plus emotional distress, such as anxiety, stress, and depression, and financial toxicity, such as insurance and job discrimination.
AS is not necessarily a picnic. You have to stay on top of PSAs, MRs, and biopsies. About half of men are anxious, and 10% are so anxious that they seek treatment they technically don’t need, as AnCan and others reported in a survey of 460 men reported last year at the American Society for Clinical Oncology.
Wouldn’t we be better off if there were options to rule out cancer risks and we don’t have to be tagged as cancer patientspatients.? Maybe AI can help.
(Dr. Kirk Wojno)
And will pathologists go redundant? Can biopsies, if they’re still be done, be analyzed automatically just as our PSAs blood levels are? In the end, human pathologists may review the results and take responsibility.
Pathologist Kirk Wojno, MD, medical director of Immunis AI, a Detroit area start-up, that is developing an AI system to help patients decide between AS and aggressive treatment, thinks it may be a few years but pathologists like him and the Gleason system are on the way out. Sure, docs may review but AI systems, using the experince of tens of millions of biopsy slides, could call the shots.
“As a pathologist, I've thought for a long time I will eventually be replaced. Hopefully, I'll be retired before I'll be replaced,” he said. “But I think these image analysis tools that I've been studying for a long time are highly trainable. The holdup now is really regulatory and the continuous calibration and validation These systems can continuously evolve and get better.”
Wojno said there are so many varieties of prostate cancers “that I think the computer eventually can do a better job than we [human pathologists] can, and it'll put it into better buckets and better predictions and again have large datasets. I can't predict how long that'll take, but I do envision computer reclassification.”
Before AI systems ultimately “take over,” Wojno said they can be used to screen cases that then are referred to a pathologist, or AI can double check the pathology report“ to make sure that the pathologist didn't miss anything. It's a great quality control tool to do it like that.”
There are unresolved liability issues for AI use. No system is perfect. Humans, especially, can make mistakes. But who takes on the responsibility, financial or otherwise, if an AI system goofs.
Spratt said he thinks pathologists will stay on the job in some capacity. “It's pathologists who refuse to use AI that will be the ones replaced. I use the analogy of a typewriter versus Microsoft Word. Microsoft Word gives us little squiggly lines under all our misspellings and auto-checks things as we go. It would be archaic to use a typewriter. We're getting to the point now with ChatGPT that it can probably write the paragraph for you, but you may want to fine-tune it.”
ASPI has posted the video for the AI program, “How AI will decrease overdiagnosis and overtreatment of prostate cancer,”at https://aspatients.org/meeting/how-ai-will-decrease-overdiagnosis-and-overtreatment-of-prostate-cancer/
Don’t forget the virtual AS session at the ZERO Summit
By Howard Wolinsky
For the past three years, I have run a special virtual Active Surveillance support group for ZERO Prostate Cancer.
Join us at 11 a.m.-12 p.m. Eastern on Tuesday, March 12, 2024. Register:
https://us02web.zoom.us/meeting/register/tZUsfuqgrjIoG9AWf7voMhzT_UjdqbQQbQPA
My stalwart AS support colleagues Jim Schraidt, a ZERO board member and from AnCan, and Hugh Idstein and Garry Tosca from AnCan, will be on deck at this popular session.
Send your questions about AS and DNA testing, urology, radiology, pathology, sexual health, and lifestyle via email to mailto:pros8canswers@gmail.com, or cut and past pros8canswers@gmail.com
Keep the questions short and sweet. They should be of general interest. Sign with your real name, or just initials, tell me where you live, how long you‘ve been on AS, are you seeing a private practice or university urologist, tell me how it’s going for you. Share a whimsical signature if you’re so inclined. Like ‘Butch from Idaho” or “Lost in Flossmoor.’)
Question: I was diagnosed with low-risk Gleason 6 five years ago. I have had a couple of biopsies and MRIs. But my urologist has never recommended a genomic/somatic test like Decipher or Prolaris. Who should have those tests? What do they do? How is DNA/germline testing different from somatic/genomic testing?
Genetically shuffling in Buffalo
Dr. Singhal answers:
All tests, including biopsies, imaging (MRI, PSMA, bone scan, CT), and labs (PSA), help us risk stratify patients with prostate cancer. Genomic tests are no different. There are a multitude of available tests for use in prostate cancer that have been developed and studied.1 Our guidelines suggest using these in situations where their results will help guide our clinical decisions. This can be in the initial screening setting, selecting who should be placed on active surveillance, identifying those who have clinically significant disease, or predicting long-term outcomes such as recurrence, metastasis, or survival. There is no recommendation for routine use of these tests, as many have not been tested prospectively in large clinical trials or been shown to actually improve long-term clinical outcomes for patients. Nonetheless, they can be helpful in specific situations where a test result may alter a provider’s decision-making.
Decipher and Prolaris are two examples of genomic tests, among many. These tests are run on samples from the tumors of patients and analyze the expression level of specific genes that are included in the assay. By understanding the expression levels of the genes included in these tests, they have the ability to predict the probability of certain outcomes (risk of metastasis, presence of clinically significant cancer, need for salvage therapy, etc). While these types of tests can be useful in certain scenarios, it is still important for providers to consider the whole picture, ie. combine all the available information (PSA, biopsy result, imaging findings, genomic test result, patient-specific characteristics), when making a clinical decision.
Germline testing, in which inherited DNA is tested, not just from the tumor sample, is recommended by guidelines for patients with many different cancers, including prostate cancer. Germline testing for prostate cancer is recommended for those with a personal history of prostate cancer in specific scenarios (Table from NCCN guidelines). It includes testing for specific genes associated with familial cancer syndromes, as those with mutations in these genes may respond better to certain therapies. Further, identifying those with these high-risk mutations can allow their family members to be screened for cancer as well.
1: Molecular Biomarkers in Localized Prostate Cancer: ASCO Guideline
Scott E. Eggener, R. Bryan Rumble, Andrew J. Armstrong, Todd M. Morgan, Tony Crispino, Philip Cornford, Theodorus van der Kwast, David J. Grignon, Alex J. Rai, Neeraj Agarwal, Eric A. Klein, Robert B. Den, and Himisha BeltranJournal of Clinical Oncology 2020 38:13, 1474-149
Udit Singhal, MD currently is a fellow in urologic oncology at the Mayo Clinic and University of Michigan, and will be joining the University of Michigan as an Assistant Professor in Urologic Oncology in July 2024. He has an interest in the management of complex urologic malignancy, including open, laparoscopic, and robotic surgical approaches for the management of kidney, prostate, bladder, testis, and penile cancers. His research interests include the development, and validation of biomarkers in urologic cancers, with a specific focus on identifying avenues for integration of biomarkers into clinical practice.
His long-term research objective is to run well-conducted biomarker-driven clinical trials in urology. He has received research funding from the National Cancer Institute, Urology Care Foundation, Prostate Cancer Foundation, American Urological Association, Howard Hughes Medical Institute, and Merck & Co, Inc. He has had many peer-reviewed publications in leading academic journals, including the New England Journal of Medicine, Journal of the American Medical Association, and Nature, among others.
Note: This blog is intended for informational purposes and not as medical advice.
Another prostate humor winner
Steve Mael, of Hudson, Wisconsin, is a winner in our Putting the Glee in Gleason Prostate Humot Contest.
As a gift, he received the book, “Riding the Scapel,” a travel/prostate cancer memoir by Jim Doilney and Rick Barrow. Steve is an inventor, tinkerer, outdoorsman, and more.
He said: “In mid-2018 I was ‘diagnosed’ with Gleason 6 and immediately sought out and attended meetings with Twin Cities - MN Prostate Cancer Support. As a result, I told three doctors that I wanted to be on AS before settling on a fourth urologist who reluctantly obliged.”
Here’s his entry: