Breaking: Urine-based MyProstateScore-AS Biomarker Hits the Marks Again, outpacing MRIs
An update on new urine test to help men with low-risk PCa skip biopsies--and MRIs
By Howard Wolinsky, Editor, The Active Surveillor
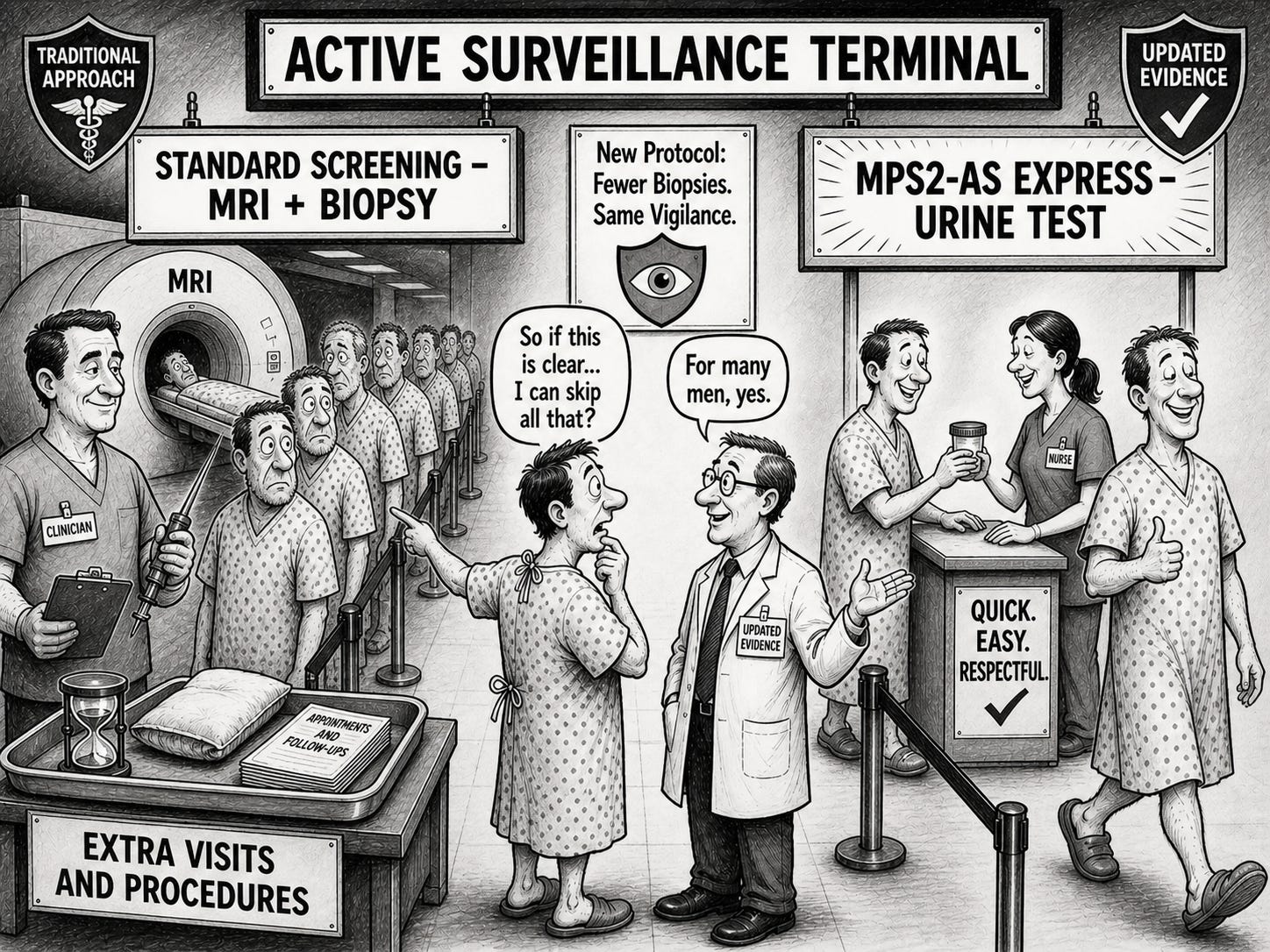
A new study suggests that many men on Active Surveillance for prostate cancer could safely skip scheduled biopsies and MRIs, as a urine test—MyProstateScore 2.0 – Active Surveillance (MPS2-AS) outperformed MRI at identifying when seemingly “low‑risk” disease has upgraded to a more aggressive form.
(Art by Perplexity AI & The Active Surveillor.)
“Fifteen years ago, Active Surveillance required annual biopsies to ensure higher-grade cancers were not missed. The goal was always to find a non-invasive approach to monitoring that did not compromise the excellent cancer outcomes achieved with yearly invasive biopsies,” lead author Jeffrey Tosoian, MD, MPH, founder of Lynx Dx, developer of My Prostate Score 2.0 and now MPS2-AS, told The Active Surveillor.
The researcher, an assistant professor in the Department of Urology, Division of Urologic Oncology at Vanderbilt University in Nashville, added:“These findings suggest that this testing approach could usher in that era of non-invasive surveillance. For patients on AS, this means an equally safe but far less invasive approach to monitoring. In other words – fewer biopsies and fewer MRIs.”
(Dr. Jeffery Tosoian)
[Note: Tosoian will be speaking in late June at the monthly webinar of Active Surveillance Patients International. Details soon.)
He added: “Moving forward, we plan to further demonstrate the accuracy of MPS2-AS in larger and more diverse cohorts. At the same time, in addition to existing tests for early detection (i.e. MPS2 for patients with elevated PSA) and accurate monitoring on surveillance (MPS2-AS), we aim to apply this urinary testing approach to improve other phases of prostate cancer care, such as early and accurate detection of recurrence after treatment.”
Ann Arbor-based Lynx Dx, the manufacturer of the test, is preparing to make it available for clinical ordering, with Medicare coverage at launch. “We will share specific availability details as launch planning is finalized,” a company spokesperson said,
The multicenter study, published in The Journal of Urology in connection with the American Urological Association meeting starting May 15, evaluated MPS2‑AS, in 330 men with Grade Group 1 prostate cancer on Active Surveillance at 11 centers.
All subjects were already slated for a protocol surveillance biopsy. Before biopsy, each man provided a urine sample, and most also underwent multiparametric MRI. Biopsies included at least 12 systematic cores, with targeted samples added when MRI showed PI‑RADS 3–5 lesions.
On biopsy, roughly 37% of men were upgraded to Grade Group 2 or higher and 9.4% to Grade Group 3 or higher—levels that often lead to curative treatment rather than continued surveillance. The paper reports that the MPS2‑AS urine test was more accurate than MRI at predicting both types of upgrading, showing stronger discrimination for who would harbor Grade Group 2+ and Grade Group 3+ disease at the time of biopsy.
A clinical decision analysis underscored the potential impact on practice.
When used as the gatekeeper for biopsy, MPS2‑AS would have avoided about 64% of biopsies later judged unnecessary while missing only around 3.2% of Grade Group 3 or higher tumors. By comparison, an MRI‑based strategy using PI‑RADS 3 or above as the biopsy trigger would have avoided fewer biopsies—about 50%—and missed many more serious cancers, with roughly 18% of Grade Group 3+ upgrades going undetected.
The test’s performance was consistent across key subgroups, including Black and non‑Black men and both confirmatory and subsequent surveillance biopsies.
Last December, The Active Surveillor presented early results on the Lynx Dx biomarker development as the potential “holy grail,” a way to monitor men with a non‑invasive urine test and reserve MRI and biopsy for those at genuine risk of progression. At that stage, much of the enthusiasm rested on early data and the biological logic of the 18‑gene panel.
Christian Pavlovich, MD, director of the Prostate Cancer Active Surveillance Program at Johns Hopkins, confirmed that no secondary biomarkers have been helpful to date in active surveillance.
He told The Active Surveillor in December, “MPS2-AS might be the biomarker we have been looking for. While not all GG2 is indolent, and any pattern 4 is potentially dangerous, there is certainly benefit to a urinary test that can help decide if and when surveillance MRI and biopsy are needed. So let’s hope MPS2-AS ushers in a new era of molecular prognostication for patients living with untreated prostate cancer. Of course, we will have to see the data in full before getting too excited, but this abstract certainly suggests there is much to be gained by using this test.”
The new study moves that idea into a more mature phase.
In a real‑world, multicenter active‑surveillance cohort, the surveillance‑specific model, MPS2‑AS, not only held up but also clearly outperformed MRI in detecting clinically important upgrading. If these findings are adopted into guidelines and practice, active surveillance could shift toward protocols that start with a urine test like MPS2‑AS and use MRI and biopsy more selectively—potentially reducing the burden of repeated invasive procedures and scans for many men, without giving up the safety of catching meaningful changes in their cancer in time.
ASPI to Host Dr. Adam B. Weiner on Living Well With AS
On Saturday, May 23, 2026, at noon Eastern, Active Surveillance Patients International will host Adam B. Weiner, MD, a urologist at Cedars-Sinai, for a program titled, “Living Well on Active Surveillance: What Matters Most (and What Doesn’t).”
REGISTER HERE: https://zoom.us/meeting/register/VfFFjirZSbyHoc8cNIMZJQ#/registration
For men on Active Surveillance, one of the continuing challenges is separating what truly deserves attention from what may only add anxiety. Weiner will discuss the key components of Active Surveillance, what patients can do to optimize their monitoring, and what is currently known about the role of diet, exercise, and supplements in prostate cancer care.
He will also address the uncertainty that can accompany Active Surveillance and offer practical guidance on what matters most — and what may not warrant as much worry.
Questions are welcome. Patients may submit them in advance to contactus@aspatients.org. There will also be a dedicated time for live Q&A following the presentation.




Again, Dr. Holden, as with your comments in recent blog, "NCCN patient guide argues that the benefits of PCa screening now outweigh the harms," thank you! I would NOT otherwise known, use off-label to garner FDA approval, Medicare and commercial approval and ultimately M O N E Y.. As many who follow this blog are silent, your participation is appreciated if rarely expressed. Again reason to show financial support for this unique blog.