Does "T" fuel the fire of cancer? More research needed on testosterone, but T not ruled out in patients on Active Surveillance
Meanwhile, Artera AI needs your help
(Send your questions about AS and urology, radiology, pathology, sexual health, and lifestyle via email to mailto:pros8canswers@gmail.com)
Question: I am 65, on AS for low-risk prostate cancer, have “low T” and low libido. Can I safely get testosterone therapy without adding “fuel to the fire”?
Dr. Leapman: The question of testosterone and active surveillance is important and comes up often.
It has long been known that prostate cancer growth is driven by androgens (male sex steroid hormones), including testosterone. Androgen suppression (androgen deprivation therapy) has been a mainstay of treatment for advanced prostate cancer for decades.
So, the conventional thinking has been to avoid external forms of testosterone in situations of known or suspected prostate cancer. As a hormone, relatively small amounts of testosterone are needed to function as a signal in the body, including in prostate cells. That is why, testosterone supplementation to very high levels is not recommended for individuals who have levels in a “normal” range.
For this reason, there is increasing comfort with the use of testosterone supplementation (“exogenous testosterone”) in patients to reach “normal” levels. In a recently published randomized trial (https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2813293) supplementing testosterone in men with low levels did not increase risks of finding high-grade prostate cancer compared with a placebo.
But what about patients with a known diagnosis of prostate cancer? Concerns that adding testosterone could serve as “fuel to the fire” have led to apprehension about supplementation. This is still a concern in patients with aggressive or metastatic prostate cancer as treatments center on decreasing testosterone levels. However, For patients who have been treated without evidence of recurrence, there is increasing comfort with administering testosterone for patients experiencing symptoms and lab levels showing low testosterone.
For patients with low-grade prostate cancer on active surveillance, the role of testosterone has also been explored. To date, there has not been a high-quality randomized trial in patients on active surveillance. However, evidence from retrospective studies suggests that testosterone supplementation was not associated with increased risks of progression or need for active treatment. https://pubmed.ncbi.nlm.nih.gov/37423815/
Although this evidence is encouraging, I would say this is still an area where stronger evidence is needed. Patients on active surveillance require monitoring to ensure that there are no significant changes over time. At the moment there does not appear to be compelling information – either from clinical studies or a strong biological rationale – suggesting that otherwise appropriate candidates for active surveillance should not be offered monitoring because of testosterone supplementation.
Michael S. Leapman, MD, MHS. is an associate professor of urology and clinical program leader, the Prostate & Urologic Cancers Program, Yale Cancer Center, New Haven. He has a special interest in low-risk prostate cancer, Active Surveillance, nerve-sparing robotic prostatectomy, focal therapy, high-risk disease, molecular imaging, and PSMA PET scans. Send Dr. Leapman questions on Active Surveillance at mailto:pros8canswers@gmail.com.
Artera needs input from low-/intermediate-risk patients on its pioneering AI technology
By Howard Wolinsky
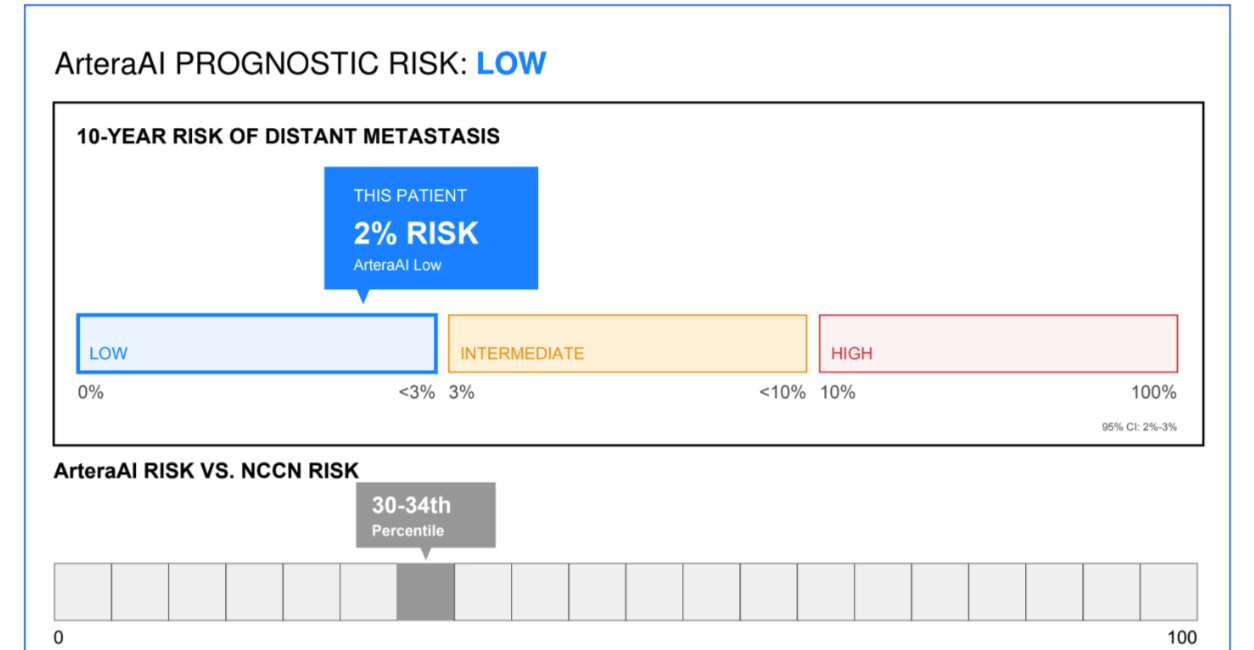
Artera, the San Francisco-based AI company, is blazing new trails in prostate cancer.
The Centers for Medicare and Medicaid Services in January cleared the way for reimbursement of the Artera AI as the first to help determine if men will benefit from Androgen Deprivation Therapy. Then, the National Comprehensive Cancer Network, which writes guidelines for cancer care, made Artera the first AI it has put into its cancer guidelines.
The test is available throughout the U.S. with exception of California and New York, which have their own approval processes.
Artera later this year plans to introduce a test to help decide whether patients will benefit from active surveillance.
Now, the startup is looking for some input from you.
Michael Chang, marketing director at Artera, told me the company is interested in gathering feedback from patients and their care partners to improve its educational and marketing materials.
“Specifically, Artera would like to hear from patients currently on active surveillance, as well as those with intermediate-risk disease that have, or, are contemplating radiation therapy with or without androgen deprivation therapy,” Chang said.
If interested, please contact michael.chang@artera.ai
Some background:
Artificial Intelligence arrives for intermediate PCa--coming soon for low-risk patients
By Howard Wolinsky Editor’s note: Artificial Intelligence is the topic of everyday discussion—at my house, anyway. I have covered AI in this blog and elsewhere for years. AI is finding its place throughout medicine, from answering our queries in portals to forecasting our risks for various disease and potential benefits for treatments.
Unsolicited testimonial
Vox story: https://www.vox.com/health/24055531/prostate-cancer-lloyd-austin
Recorded interview with Noel King of Vox/NPR: https://shorturl.at/bpxz1
I also appeared on the “Stay in the Game” podcast from Fans for the Cure. Give it a spin: https://fansforthecure.org/podcast/the-active-surveillor-when-the-best-treatement-is-no-treatment
More is coming soon.





God advice. The Dude abides.
AI? Hmmmmmm best be VERY careful!
https://secularheretic.substack.com/p/children-playing-with-bombs?publication_id=226642&post_id=143333761&isFreemail=true&r=18qe84&triedRedirect=true